Car T Cells Ema
The european medicines agency ema has recommended the first two marketing authorisations for chimeric antigen receptors car t cells medicines in the european union eu.
Car t cells ema. The marketing authorization application submitted by bristol myers squibb is specific to patients with relapsed or refractory diffuse large b cell lymphoma dlbcl primary mediastinal b cell lymphoma pmbcl and follicular lymphoma grade 3b fl3b who have tried at least two prior therapies. They belong to a new generation of personalised cancer immunotherapies that are based on collecting and modifying patients own immune cells to treat their cancer. C15a3 cea 0. Martina schüssler lenz germany is the chair of the ema committee for advanced therapies cat.
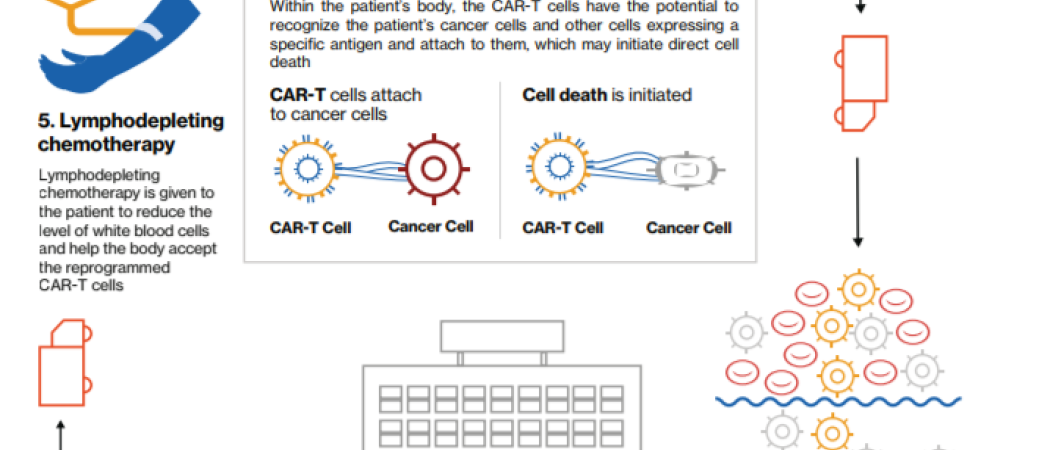
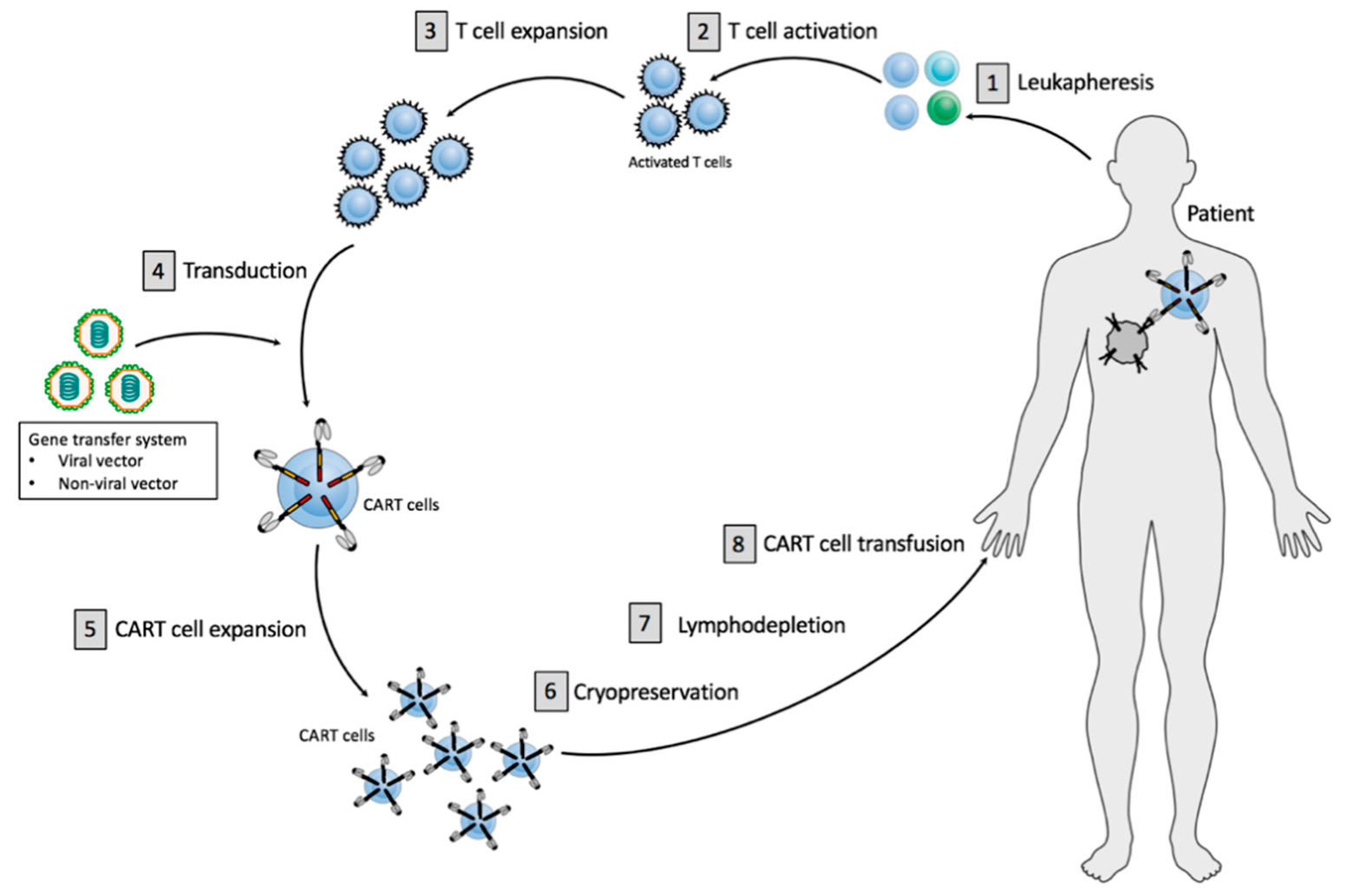
Gene transfer technology is used to stably express cars on t cells conferring novel antigen specificity1 2 ctl019 therapy takes advantage of the cytotoxic potential of t cells thereby killing tumor cells in an antigen dependent manner1 3 persistent ctl019 cells consist of both effector cytotoxic and central memory t cells3 1. The recommendations in the report are based on the chimeric antigen receptor car t cell therapy registries workshop organised by the ema on february 9th 2018 to which the ebmt was invited along with cibmtr as part of the european agency s patient registry initiative and stakeholders from industry professional societies patient representatives health technology assessment agencies and national regulators. In her presentation she explained how the cat operates. The cat is responsible for assessing the quality safety and efficacy of atmps.
T cells w o t cells car t cells car iil 12 c15a3 cea tumor volume mm 3 a b c t cells engineered with car inducible il 12 0 500 1000 1500 2000 0 20 40 60 0 500 1000 1500 2000 0 20 40 60 0 500 1000 1500 2000 0 20 40 60 days after tumor inoculation t cells car iil 12 irrad. 293t cells cil 12 t cells car iil 12 irrad.